 [8 MIN READ]
[8 MIN READ]
We are all fallible. Despite their best efforts, physicians, nurses, other healthcare workers, and indeed all humans, make errors. It is an essential component of our being. It is widely accepted that a systems approach to error prevention can counteract this fallibility. Nonetheless, errors do occur. These failures can often be explained by the “Swiss Cheese Model” of error. Multiple processes are designed to prevent a type of error. Each individual process may be imperfect but used in combination one process will succeed where another may fail. Like the holes in Swiss cheese, when these imperfections are aligned and multiple processes fail, errors occur.
Below I present three cases from which we can gain perspective on how poor communication, systems errors, and cognitive bias contribute to surgical errors.
Retained Foreign Body
CASE
An elderly gentleman presented with severe claudication and absent femoral pulses. Angiography showed severe aortoiliac occlusive disease, and he was taken to the operating room for an aortofemoral bypass. A midline abdominal incision was made and after abdominal exploration the aorta was exposed using laparotomy pads and a self-retaining retractor. The common femoral arteries were exposed and surrounded with vessel loops. After administration of heparin anticoagulation, a 14x7mm knitted Dacron aortofemoral graft was placed with bilateral onlay profundaplasties. Anticoagulation was reversed and bleeding was controlled. The retroperitoneum was closed.
All laparotomy pads, sponges, vessel loops and instruments were then removed, and the abdominal contents returned to their normal anatomic position. Towel, laparotomy pad, sponge, needle and instrument counts were requested by the attending surgeon and were reported as correct. A manual examination (“sweep”) of the abdominal cavity was performed and there were no retained foreign bodies. The fascia was closed with interrupted sutures and skin closure was accomplished with staples. A comprehensive final count was correct. During extubation the patient developed a coughing paroxysm and the wound dehisced with subsequent evisceration of small bowel. The bowel was controlled with moist laps, the patient was re-intubated, and the wound closed.
Postoperatively the patient did well but developed a persistent ileus with poor bowel sounds and abdominal discomfort. He became distended, despite a nasogastric tube and an abdominal radiograph showed distended loops of small bowel and the radio-opaque marking of a laparotomy pad. He was returned to the operating room where a single laparotomy pad was recovered. The remainder of his postoperative course was uneventful. On long-term follow-up he continued to do well with palpable pedal pulses and no evidence of infection.
ANALYSIS
Although appropriate policies and procedures were followed during the initial procedure, the team in the operating room failed to maintain their composure when the patient eviscerated. First, no counts were performed prior to or after the secondary wound closure. Second, the abdominal cavity was not manually explored prior to closure. Third, had these two lapses in protocol been recognized, a foreign body film could have been taken in the operating room. The failure to count, the failure to explore and the failure to identify these deviations from standards aligned to result in a retained foreign body.
Inadvertent Anticoagulation
CASE
 An 82-year-old male presented with abdominal pain and distention. He was hypotensive with a systolic blood pressure < 80 mmHg. He was taken to the operating room with a diagnosis of a ruptured abdominal aortic aneurysm (AAA). The aorta was rapidly exposed and clamped just below the level of the renal arteries. Distal control was obtained at the iliac level and an 18mm woven Dacron aortic tube graft was placed. The clamp was removed as additional volume replacement was administered to maintain a satisfactory blood pressure.
An 82-year-old male presented with abdominal pain and distention. He was hypotensive with a systolic blood pressure < 80 mmHg. He was taken to the operating room with a diagnosis of a ruptured abdominal aortic aneurysm (AAA). The aorta was rapidly exposed and clamped just below the level of the renal arteries. Distal control was obtained at the iliac level and an 18mm woven Dacron aortic tube graft was placed. The clamp was removed as additional volume replacement was administered to maintain a satisfactory blood pressure.
As closure was begun, the onset of diffuse bleeding was noted. The anesthesiology team, which had changed during placement of the aortic graft, was advised of the bleeding and the possibility of a transfusion reaction or a consumption coagulopathy was raised. The anesthesiologist remarked that there should not be any bleeding since he had already administered a full dose of Protamine to reverse the heparin. The attending surgeon advised the anesthesiologist that heparin was rarely if ever administered during repair of a ruptured AAA and had not been requested, nor given, in this case. He therefore requested that heparin be administered to bind the protamine.
The diffuse bleeding resolved within minutes and the remainder of the procedure was uneventful. The patient’s postoperative course and long term follow-up were unremarkable.
ANALYSIS
Several breakdowns in standard procedure led to this, fortunately reversible, error. Although heparin use in patients with ruptured AAA remains controversial, in this institution and currently it was routine to perform surgery for a ruptured AAA without the use of heparin. Ideally, anesthesia staff should have been aware of this routine. In addition, a “shift change” in anesthesiology staff during the most critical portion of the procedure is questionable at best.
These factors aside, the written anesthesia record, including all drugs administered, maintained throughout the procedure is a systems approach to patient management. First, the anesthesiologist should have referred to the anesthesia record to determine if heparin had been administered and if so in what dose. This was not done. Second, there was a clear failure of communication. Close cooperation and coordination between the surgeon and the anesthesiologist, especially during the clamping and unclamping of the aorta, is essential. The anesthesiologist should have communicated with the surgeon to determine if, when and how much Protamine should be administered. This was not done. The anesthesiologist’s comment that the Protamine had been administered was fortuitous but led to the recognition and resolution of the problem.
Diagnostic Error
CASE
A thin middle-aged female presented to the Emergency Department (ED) with a history of sudden lower extremity paresis and paraesthesia of several hours duration. She had no history of sensory or motor deficits and no history of claudication. Past medical history was significant for a cardiac valve replacement after which she was placed on Coumadin anticoagulation. She was variably compliant with medication and had not taken Coumadin as prescribed.
The possibility of a spinal cord lesion was considered, and a neurosurgical consult was requested. The patient was seen by a general neurosurgical resident and admitted to the neurosurgical service. Shortly after admission her case was discussed among the surgical residents and the possibility that her paresis and paraesthesia could be vascular in etiology was raised by a general surgery resident. A Vascular Surgical consultation was obtained, and she was seen by the attending vascular surgeon.
On physical examination there were no cervical or subclavian bruits and, in the supine position, no audible cardiac murmur. Upper extremity pulses were normal. The aortic pulse was palpable and not enlarged. Femoral and distal pulses were absent and both lower extremities were somewhat pale and cool to the touch. There was bilateral lower extremity paresis and paraesthesia but no paralysis or areas of anesthesia. A diagnosis of aortic or bilateral iliac occlusion was made and the patient was taken immediately to the Operating Room.
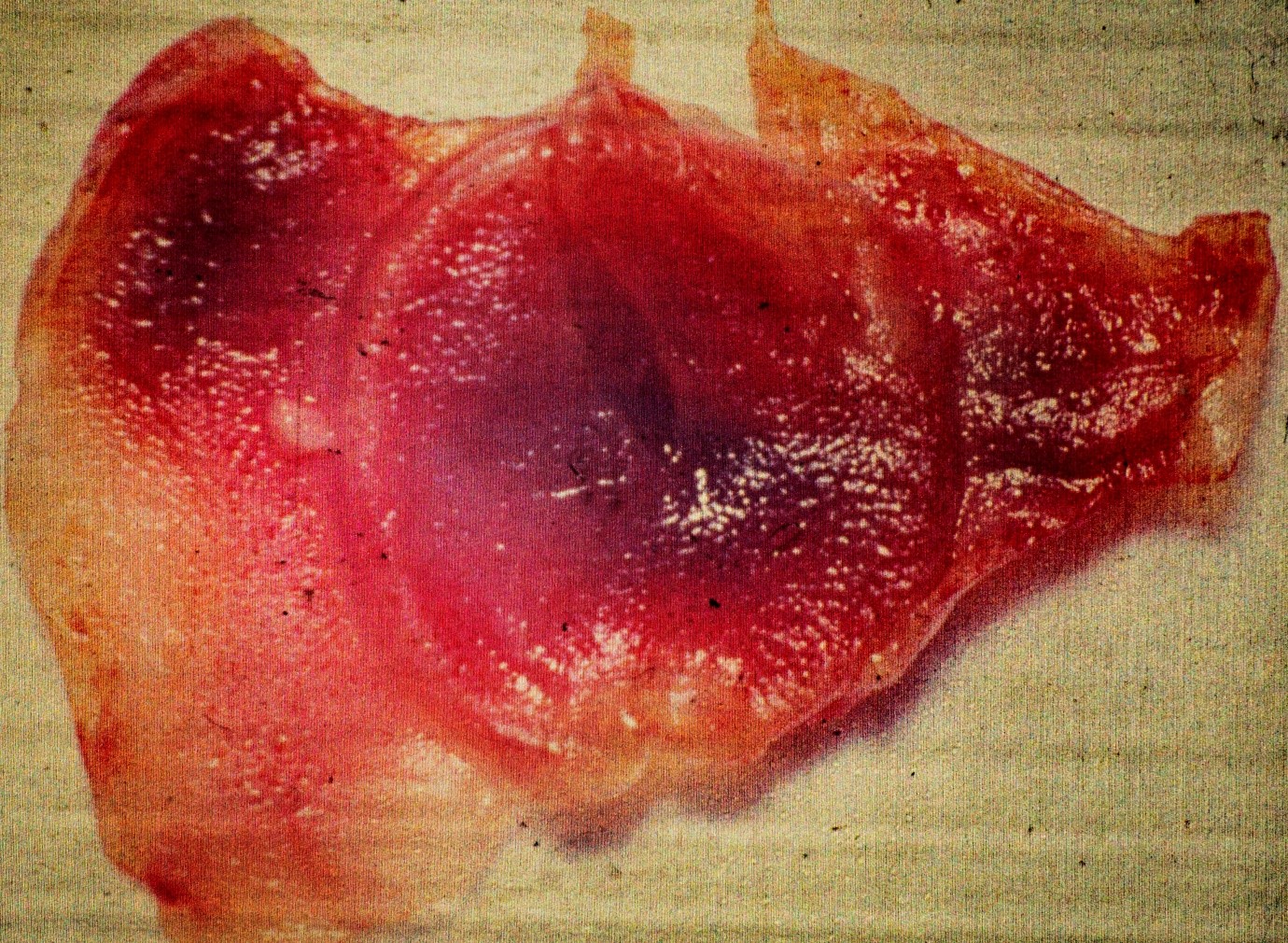
Systemic anticoagulation was obtained with intravenous heparin administration. Both femoral arteries were exposed proximal and distal control was obtained. With the left femoral artery occluded, the right femoral artery was explored through a transverse arteriotomy. There was minimal plaque and no thrombus at this level. Antegrade flow was severely reduced as was retrograde flow. A # 4 Fogarty Embolectomy catheter was passed proximally into the distal aorta, gently inflated with saline and withdrawn. A large fibrous embolism was retrieved intact. Upon examination of the fibrous material it appeared to be a fibrous cast of a cardiac valve (Fig 1). The proximal femoral artery was flushed and there was now excellent pulsatile antegrade flow. The left femoral artery was then explored. Excellent pulsatile antegrade flow was noted. No additional clots or fibrous material was obtained from either femoral artery, proximally or distally.
The arteriotomies were closed and flow was restored to both lower extremities. Femoral and distal pulses were excellent to the pedal level. Her postoperative course was unremarkable and sensory and motor functions were, and remained, entirely normal. Coumadin anticoagulation was initiated, and the importance of compliance was stressed. She remained asymptomatic on long term follow-up with palpable pedal pulses.
ANALYSIS
Most errors in surgical care occur preoperatively or postoperatively. In this case there were several process errors. First, the patient should have been seen by a qualified ED physician rather than being triaged to neurosurgery. Second and most notably, the evaluation by neurosurgery may have been flawed by cognition bias and the diagnosis of vascular embolism not considered. Finally, the absence of femoral pulses should have resulted in a prompt vascular surgical consult.
Cognition bias is well recognized as a cause of diagnostic error. There are several types of cognition error[1] which may be operable alone or in combination. Availability heuristic is making a diagnosis biased by experience and hence expectations. The presenting symptoms in this case may have been like previous cases the neurosurgical resident had seen. Alternatively, this may be an example of a “framing effect”, bias due to what the resident may have expected in a patient referred to neurosurgery. Anchoring heuristic, also termed cognitive bias or premature closure is when the initial diagnostic impression is relied upon despite subsequent information to the contrary. Here, the initial impression of a neurologic etiology resulted in an admitting diagnosis may have prevented consideration of other possibilities (premature closure).
It is not possible to determine what form(s) of cognitive bias (if any) may have influenced the initial diagnosis. Fortunately, the error was recognized, the proper diagnosis was reached, prompt operative intervention was accomplished, the patient was successfully treated and had an excellent result.
SUMMARY
Medical errors are best prevented using multiple process measures to prevent human error. However, even well-designed process measures are imperfect as they are dependent upon execution by human healthcare workers. When the imperfections of multiple processes align, like holes in Swiss cheese, errors may occur.
References
[1] Patient Safety Primer, Last Updated January 2019. Patient Safety Network of the Agency for Healthcare Research and Quality. Available at https://psnet.ahrq.gov/primers/primer/12 (Last Accessed August 27, 2019)


