[12 MIN READ]
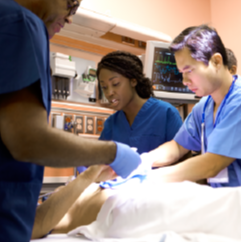
While the mission of clinical research is to advance the practice of medicine, it is an extremely daunting task to not only manage clinical trials, but to also oversee them to keep patients safe and reduce risk. In a review of numerous malpractice cases related to clinical research, I have found several common denominators and opportunities to improve patient safety.
The issues that arise from clinical trials are not typically intentional; they are simply intrinsic to the complexities of the system. Under the best of circumstances and in many cases by design, patients have significant problems and are randomized to receive a trial drug. This population is inherently at risk. Additionally, many medical professionals are not trained to conduct appropriate, safe, well-organized clinical trials. These factors along with others I will outline have led to an increase in patient injury, federal and legal scrutiny, and obviously, litigation.




.png)